Dr. Larry Istrail is a physician and author of The POCUS Manifesto: Expanding the Limits of our Physical Exam with Point-of-Care-Ultrasound (POCUS). You can get a copy here.
—
Amidst the rising carnage inflicted by the COVID-19 pandemic sat innovative scientist turned biotech mogul Dr. Jonathan Rothberg, from his yacht. On March 7, 2020, as the United States daily positive counts were still only recorded at 117 due to our extreme testing shortage, he snapped a photo of his clean, organized desk floating on the Atlantic ocean and tweeted the following:


At the time, the testing situation in the United States was suboptimal, to say the least. The CDC had infamously fumbled with their own test while not allowing other labs to develop one. On February 28th, 2020 Science.org published an article explaining the rest of the world’s exponential headstart:
The World Health Organization (WHO) has shipped testing kits to 57 countries. China had five commercial tests on the market 1 month ago and can now do up to 1.6 million tests a week; South Korea has tested 65,000 people so far. The U. S. Centers for Disease Control and Prevention (CDC), in contrast, has done only 459 tests since the epidemic began. The rollout of a CDC-designed test kit to state and local labs has become a fiasco because it contained a faulty reagent. Labs around the country eager to test more suspected cases—and test them faster—have been unable to do so. No commercial or state labs have the approval to use their own tests.
Just one day later, the CDC reversed course and allowed independent labs to develop their own, but with was so much unchecked exponential growth at the time, the test-and-trace levees had already been breached. By March 1st, the United States had completed just hundreds of tests throughout the entire country while South Korea, for comparison, had already done 98,000.
If anyone could develop a new COVID test from a yacht, it’s Rothberg. A biomedical engineer by training, he started multiple genomics companies, was awarded the National Medal of Technology and Innovation by President Obama, founded the Butterfly Network that created the first hand-held head-to-toe ultrasound that connects to a smartphone, and Hyperfine, a completely portable MRI machine. So his ambitious vision of an at home, highly accurate rapid test was an inspirational light at the end of the dark, 2020 tunnel. And inn just 1.5 short years, this vision would become Detect.

Polymerase chain reaction (PCR) tests: look for the genetic material of the virus
A PCR test is considered the gold standard in viral detection. It is a highly sensitive test, which is its strength but can also be a weakness. It is traditionally performed in a lab with large, expensive machines, but Rothberg wanted to turn it into a low-cost home test.
Instead of the standard PCR techniques, Detect uses a similar technique called RT-LAMP (Reverse Transcription Loop-mediated Isothermal Amplification) and lateral flow strip technologies to detect nucleic acids from the SARS-CoV-2 genome.

First, the DNA is broken apart at an elevated temperature in a disposable tube. It is then amplified millions of times, after which the tube is inserted into the reader. At the end of this process, the liquid enters the lateral flow strip for detection. Overall it takes about 1 hour.
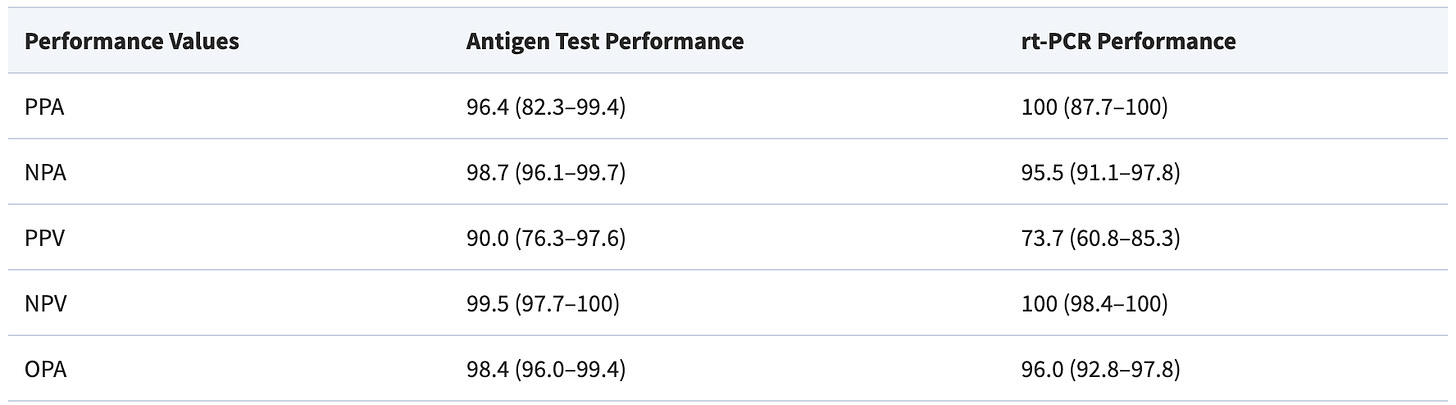
For FDA approval they submitted a clinical study where adults who had symptoms or were exposed self swabbed their nostrils and used the Detect test. This was compared to the results of a gold-standard PCR test in a lab, and the accuracy was quite impressive:
The test was 90.9% sensitive or picked up 91/100 patients who had a SARS-CoV-2 infection
It was 100% specific, meaning it only turned positive when the patient was infected, correctly identifying a negative samples every time
It was able to detect less than 1 copy per microliter, which is on par with PCR
Incredibly impressive for a home test, and especially good at detecting an infected patient early on. However, like with PCR tests after a SARS-CoV2 infection, it may remain positive long after you are infectious.
Rapid Antigen Tests: look for proteins on the surface of the virus
The push for rapid tests in the United States has been spearheaded by a Buddhist monk turned epidemiologist turned physician named Dr. Michael Mina, who has been screaming from the Twitter rooftops promoting wide-spread rapid testing since the start of the pandemic. The rapid tests - which are now available over the counter in the U.S. (atleast before they all sold out during Omicron) - take about 15-30 minutes and look for the SARS-CoV-2 nucleocapsid protein, the most abundant protein expressed by the virus using a method called lateral flow. When compared with PCR, they are about 30% less sensitive (give false-negative results 30% more) for detecting the virus.
However, this is not necessarily a bad thing.
Since PCR is considered the gold standard, it is what the rapid tests are compared to. But ideally, the gold standard should be ‘infectious person,’ not ‘person with un-transmissible dead viral particles in the nose.’
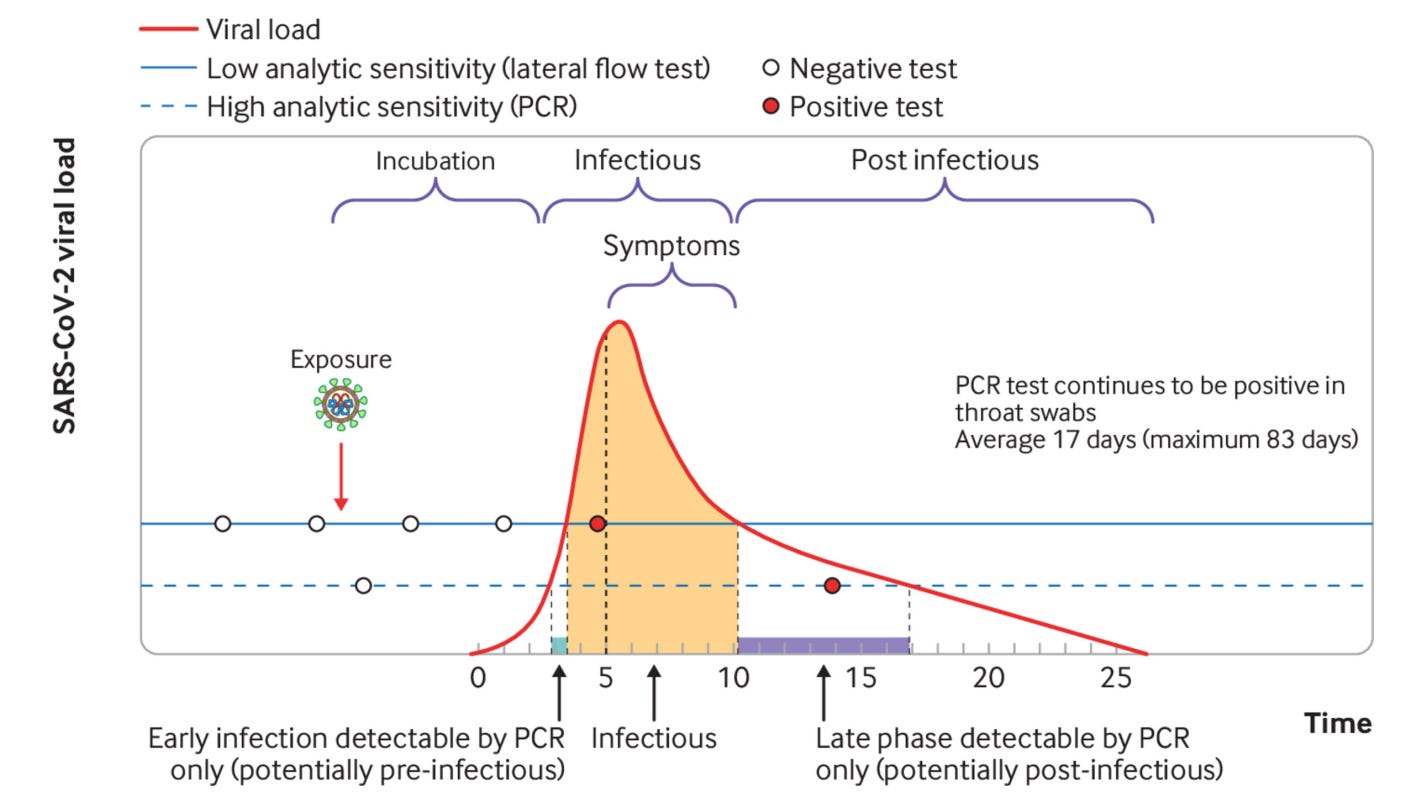
As you can see in the figure above, once a person has been infected, it takes a few days for the viral load to rise to levels that cause symptoms. Once the viral load passes a certain threshold, the rapid antigen tests (solid blue line) can detect the virus with high accuracy, at the time when the patient is most infectious. Once the infectious period ends, and the viral load reduces, the rapid test can no longer detect it, while the PCR can remain positive for an average of 17 days, but can be up to 83 days or more.
So while the rapid test will miss some people early on in their infection (give a false negative) that a PCR test would detect, it will not give a false positive result weeks after the patient’s infectious period. In this regard, it is highly sensitive and specific for high viral load, which is why Dr. Mina refers to it as the “contagiousness test.”
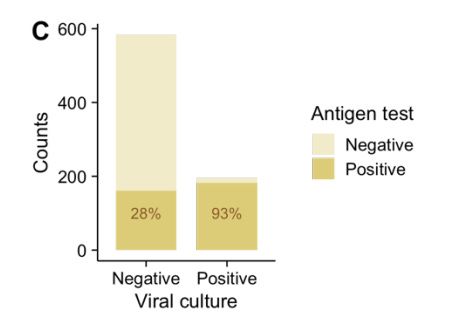
For example, in a study comparing rapid tests with PCR for their ability to detect virus that can grow in a lab (and therefore presumably be transmissible to another person), the rapid tests actually performed better. The ability of a positive rapid test to predict culture growth was 90%, compared to 73% with the PCR. The ability of a negative rapid test to predict no culture growth was 99.5% with the rapid test and 100% with the PCR.
Similar results were seen in another comparative study of rapid tests and PCR, as well as in a study using lab culture positivity as the gold standard. If the viral culture can grow in the lab, this suggests the viral load is high and the patient is very contagious. In patients with virus that grew in culture, the antigen tests picked up 93% of them:
So the limitation to rapid tests is they may miss an infectious person early on in their symptoms before their viral load reaches a detectable level, which could have serious consequences in a pandemic, or at grandma’s birthday party. But on the plus side, as the Infectious Disease Society of America phrases it, “a major advantage of these assays is the lower likelihood of detecting residual viral nucleic acid leftover from a remote infection in recovered individuals.”
This sentiment was echoed by Dr. Mina and colleagues in a New England Journal of Medicine article:
“[rapid tests] do not have an amplification step, so their analytic limits of detection are 100 or 1000 times higher than that of the benchmark test, but that is largely inconsequential if the goal is to identify people who are currently transmitting virus. SARS-CoV-2 is a virus that grows quickly inside the body, so by the time a benchmark PCR test becomes positive, the virus is well into exponential growth. At that point, it is probably hours, not days, before the virus grows by orders of magnitude, reaching the detection thresholds of currently available cheap and rapid point-of-care tests. It is after this point, when people would have positive results on both tests, that they would be expected to become infectious.”
This was also seen in a 3,300 person study comparing the now available Binax Now rapid antigen test to PCR. In patients with high viral load (Ct count of 30 or less), the Binax now tests were 100% sensitive and 98% specific. The researchers concluded that the Binax NOW “reliably detects persons with low Ct, correlating to high viral load. Rapid tests may miss individuals at the earliest rise in virus levels, a limitation that can be addressed through repeat rapid testing. Rapid tests may also miss the latter end of the viral dynamic curve (which can last for weeks), a period during which virus levels are low and a person is not thought to be infectious; some have suggested the lower sensitivity of the assay during this period could reduce hardship resulting from unnecessary isolation.”
Concern with Omicron
Some have raised concerns that the rapid tests may not work as well with the Omicron variant. Since it is so contagious, it may be more transmissable with a lower viral load before it reaches the rapid antigen test threshold. A study tracked 30 fully vaccinated people with daily rapid antigen test and PCR tests in December 2021, and found exactly what we may expect for a more contagious virus:
Day 0 to Day 1: PCR positive but all rapid antigen tests were negative. This despite a relatively high viral load with Ct <29.
Median time for rapid antigen test to become positive was 3 days
So in these cases, a rapid only approach would have missed multiple patients who can transmit the virus.
Optimal testing strategy
The CDC recently announced they are reducing the isolation time to 5 days with no recommendation for a negative rapid test. Here is Dr. Walensky discussing it on CNN:
CNN Correspondent: “Why didn’t the CDC recommend taking a rapid test after those five days?”
Walensky: “…we actually don’t know how well our rapid tests perform and how well they predict if you are transmissable during the end of disease.”
Yet as NY Times columnist Zeynep Tufekci pointed out, if Dr. Walensky were to consult herself from an article in 2020, her answer may have been different. At that time she argued that in a pandemic, rapid tests are actually better than PCR swabs. As she explained:
The more virus in the airway, the greater the likelihood of a positive antigen result. What that means is that—conveniently—there is a high degree of overlap between the period of maximal infectiousness and the period during which the antigen-based test is most likely to yield positive results. And, just as importantly, antigen-based testing is highly specific: unlike its PCR counterpart, the antigen test isn’t fooled by faint signals of lingering viral matter outside the period of infectiousness … for purposes of surveillance screening, those antigen-based negatives worrying the FDA aren’t false negatives at all; those are true negatives for disease transmission. Far from being problematic, in the context of outbreak containment, the antigen test’s limited window of sensitivity is a major asset. The antigen test is ideally suited to yield positive results precisely when the infected individual is maximally infectious
So based on what we have learned, and with the strength and limitations of PCR and rapid tests in mind, a more strategic overall testing philosophy (in an ideal world) may be:
PCR should be used for all suspected acutely infected patients. If the patient is symptomatic and PCR is positive - no further testing to confirm the diagnosis
If the patient is asymptomatic and PCR positive - check rapid test - if negative, the patient is either in initial ascent or long-tail descent - recheck rapid in 1-2 days and if negative, likely they are in the long-tail descent and not infectious
If the patient has already been symptomatic, rapid tests may be the most useful for determining when the patient is likely no longer infectious, while PCR will not be helpful in this case
Modern-day population-based monitoring
While we cannot prevent new variants of SARS-CoV2 or the next pandemic, we can use clever technologies for early detection and implement highly localized mitigation strategies.
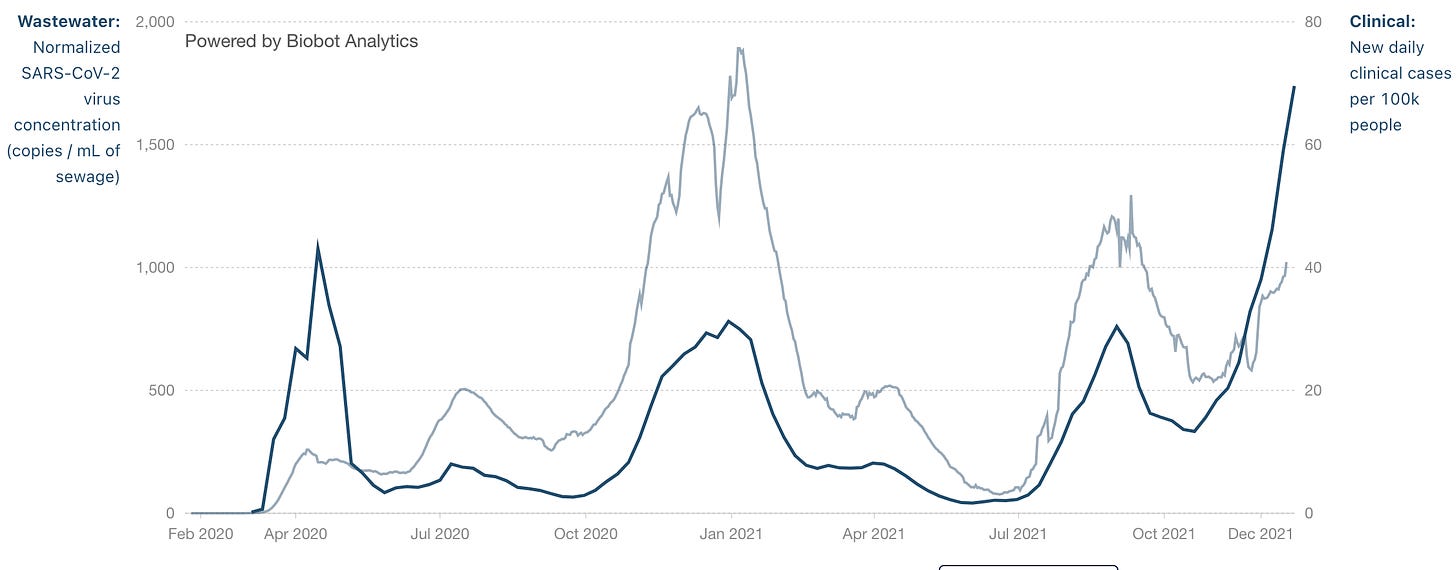
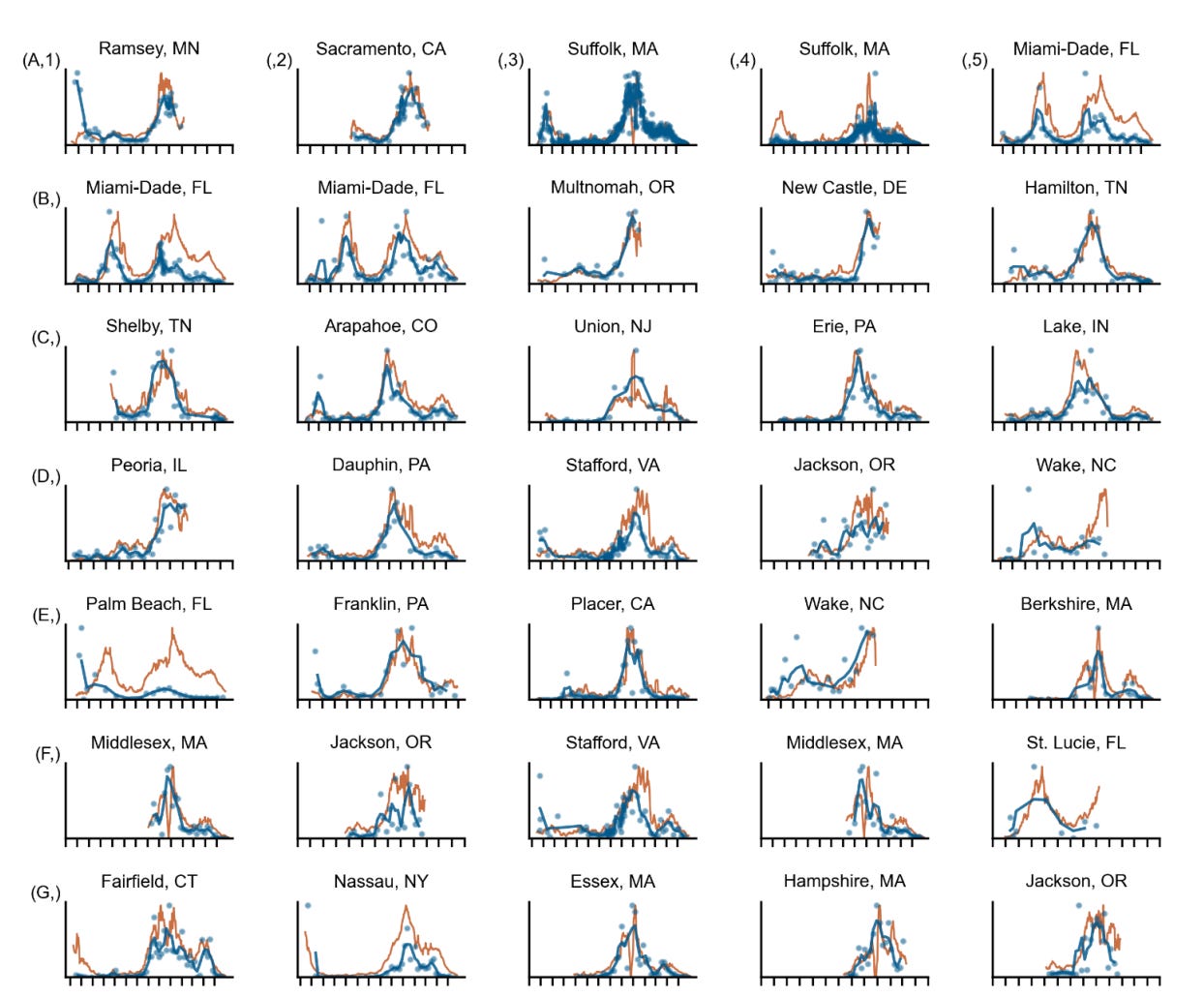
Biobot Analytics offers a unique method for surveiling COVID in the population: Sewer water.
Taking samples from sewage made up from 2.25 million individuals in Massachusetts, Biobot Analytics was able to predict outbreaks 4-10 days before they are noticed with testing in one study. And their viral levels track with the clinical testing remarkably well:
And at the state level:
Another is the Kinsa WeatherMap that uses a large network of connected thermometers mixed with COVID case data to show real-time outbreaks.
With these amazing tools, lets hope this wave peaks quickly and we can finally reach some level of normalcy again.
—
Dr. Larry Istrail is a physician and author of The POCUS Manifesto: Expanding the Limits of our Physical Exam with Point-of-Care-Ultrasound (POCUS). You can get a copy here.