Cardiac Point-Of-Care Ultrasound Origins
How cardiac ultrasound evolved into the powerful bedside tool it is today
The following is an expanded excerpt from The POCUS Manifesto: Expanding the Limits of our Physical Exam with Point-of-care Ultrasound (POCUS). You can buy it on Amazon here.
“Future generations of doctors will find it hard to believe that, in 2013, many clinicians were still relying on the vague findings of a 200-year-old traditional physical examination and were compromising clinical efficacy when direct information was available from point-of-care echocardiography. History will undoubtedly show that point-of-care echocardiography was the beginning of a ‘new glorious age’ of the physical examination.”
- Cardiologist & Cardiac POCUS pioneer Dr. Jos Roelandt, 2013
As the field of echocardiography was developing among cardiologists, thanks to the likes of Dr. Harvey Feigenbaum and others, the visual study of the heart was clearly offering more detailed and accurate information than auscultation could. With rapid advancements improving ultrasound technology and miniaturizing the hardware it required, various physicians envisioned pocket-sized devices that could view the heart as routinely as we listen to it. Dr. Jos Roelandt was one such cardiologist from the Netherlands who integrated POCUS into his physical exams as early as 1978.
Unlike most of his peers, Roelandt’s contrarian view on our collective declining physical exam abilities was decades ahead of his time. While most shunned this devolution as a negative consequence of improved imaging technology, Roelandt saw it as an opportunity. “Rather than blaming echocardiography,” he wrote in an opinion piece in the European Heart Journal in 2014, “there are many arguments why pocket-size ultrasound imagers should be used as an integral part of the physical examination at the point of care. Just like the introduction of the aural stethoscope initiated to the ‘golden era’ of clinical diagnosis, the incorporation of bedside ultrasound imaging will undoubtedly lead to a ‘new golden era’ of physical examination by providing a more accurate diagnosis, cost reduction by avoiding inappropriate tests, and a better patient–doctor rapport.” He recognized the limitations of our physical exam to detect things like early heart failure or silent valvular disease that are “a challenge to the most experienced clinician or even impossible, while they are rapidly diagnosed by echo.” Why then, he wondered, “should we rely on indirect information and secondary acoustic events when we actually can see what is wrong with the heart and its structures?”
After completing a medical degree from Belguim’s University of Leuven in 1964, he joined the Thoraxcentre in Rotterdam in 1969 and began research in clinical echocardiography.
In the mid-1970s, he and his colleagues constructed a hand-held “battery-powered ultrasound imager” they named “the ultrasound stethoscope,” that he used in his patient examinations. He was able to diagnose pericardial effusions, estimate cardiac chamber size, and look for valvular disease. They published their findings in 1978, describing their new diagnostic device. Roelandt felt the most important characteristics were “its small size, full portability and battery operation combined with a real-time imaging technique based on the linear array principle.”
In a follow-up paper, Roelandt described his initial clinical experience. In 19 of the 20 patients on the cardiology ward they scanned, “it was possible to assess the relative size of both the right and left ventricles and to study the motion pattern of the aortic and mitral valves … moderate pericardial effusion was confirmed in two patients and diagnosed in one patient in whom it was unsuspected.” On the internal medicine wards, they also found it quite useful suggesting it could “be considered as an ‘extended palpation.’”
“It is expected,” Roelandt concluded, “that this miniaturized and automated instrument will have an important impact on the diagnostic use of ultrasound and the further development of ultrasonic equipment.”
Diagnosing Heart Failure at the bedside
Since the days of Drs. Laennec and Hope, the signs and symptoms of congestive heart failure and its downstream ramifications have been well understood. Laennec knew there was a “habitually swelled state of the veins,” and Dr. Hope understood that fluid backs up into the lungs and into the rest of the body through “a series of striking phenomena,” starting with lower extremity swelling that “gradually ascends, and under the name of anasarca, may eventually attain the utmost degree over the whole surface of the body.” However, since these signs or symptoms are not specific to congestive heart failure, the direct diagnosis of a reduced ejection fraction has remained elusive.
In the Before POCUS era, listening for a third heart sound, or S3, is another sign that has been employed toward this evasive diagnosis. It was initially described by French physician Dr. Pierre Potain in 1847 as “three sounds, namely: two normal sounds of the heart and a superadded sound ... this is the bruit de galop,” becoming the subject of much scientific inquiry ever since. This third heart sound is considered normal when heard in younger patients, but after 40 years of age it is pathologic, a sign that is quite specific though not very sensitive. The diagnostic accuracies vary wildly in the literature, with sensitivities of 26 - 69%, while a review in the Journal of the American Medical Association placed the sensitivity and specificity at 13% and 99% respectively.
In more recent studies, however, this third heart sound was less specific. For example, in a study of 580 patients from 2001, it was only 45% sensitive and 87% specific for patients with congestive heart failure. Likewise, in a study of cardiologists’ auscultation abilities, the detection of an S3 was 68% sensitive and 73% specific for heart failure, while in a 2014 study of over 1300 patients with congestive heart failure and reduced ejection fraction, cardiologists only heard an S3 in 15% of them. In addition to its poor accuracy, the ability to detect a third heart sound is confounded by the fact that it is a very low-frequency sound and therefore difficult to confidently hear, and shows poor agreement regarding its existence when different physicians examine the same patient.
Seeing is Believing
This prospect of diagnosing congestive heart failure with certainty at the bedside would dramatically improve our physical examination and expedite care, instead of relying on an echocardiogram which can take hours to weeks depending on the clinical setting. In the POCUS Era, we do not need to rely on what Roelandt called “secondary acoustic events” as there are much more precise methods for determining if the patient has a reduced ejection fraction. By employing a simplified, qualitative method borrowed from echocardiography, the ejection fraction can be estimated with cardiac POCUS as normal, reduced, or severely reduced. This “eyeball method” can be accurately achieved by non cardiologists, and improve the diagnostic accuracy in a patient with congestive heart failure. Such results were evident in a study of medical students and residents from 2012, for example. Using just signs and symptoms of congestive heart failure acquired through a traditional physical exam, they detected a reduced ejection fraction with 25% sensitivity and 84% specificity. However, with the addition of cardiac POCUS and only two hours of training these figures rose to 74% and 93.6% respectively.
The capability of our physical exam to diagnose cardiovascular disease has been scrutinized in various stethoscope-versus-POCUS studies, invariably showing improved diagnostic accuracy with POCUS. In a 2001 study out of the University of Chicago, cardiologists with a stethoscope failed to detect 59% of known cardiac pathologies compared to the POCUS group who missed 29%. If only major cardiovascular findings were considered, cardiologists missed 43% while the POCUS group missed only 21%.
With the stethoscope, they missed 57% of mitral regurgitation murmurs and 76% of tricuspid regurgitation murmurs. They did well with aortic stenosis, detecting 88%, however they were particularly bad with diastolic murmurs, only detecting 26%. Two-thirds of the cardiologists could not identify patients with reduced ejection fraction with physical exam alone, while only 23% were missed with POCUS. Overall the diagnostic accuracy with POCUS was superior to the stethoscope in almost every cardiac pathology, and the authors concluded that although “cardiac auscultation has excellent specificity, the sensitivity of this technique in the current era is remarkably low.”
In a more recent study with modernized, pocket-sized ultrasound devices, the physical exams performed by 17 different cardiologists with a stethoscope were compared to the results with cardiac POCUS. Overall, the cardiologists detected only 47% of all abnormalities while 82% were detected with POCUS. The bedside ultrasound diagnosed 71% of moderate-to-severe valvular diseases compared to 31% in the physical exam group. 96% of patients with reduced ejection fraction were accurately diagnosed with POCUS compared to 35% with the exam and stethoscope (Table 5). The handheld ultrasound group was universally superior to the physical examination group, leading the authors to declare that “a physician could have an all-purpose tool in his or her pocket” that provides “timely and more accurate diagnosis than physical examination for the majority of common cardiovascular abnormalities.” It is a bedside diagnostic tool “that would be more in keeping with the 21st century than the stethoscope, a 200-year-old technology whose time should be over.”
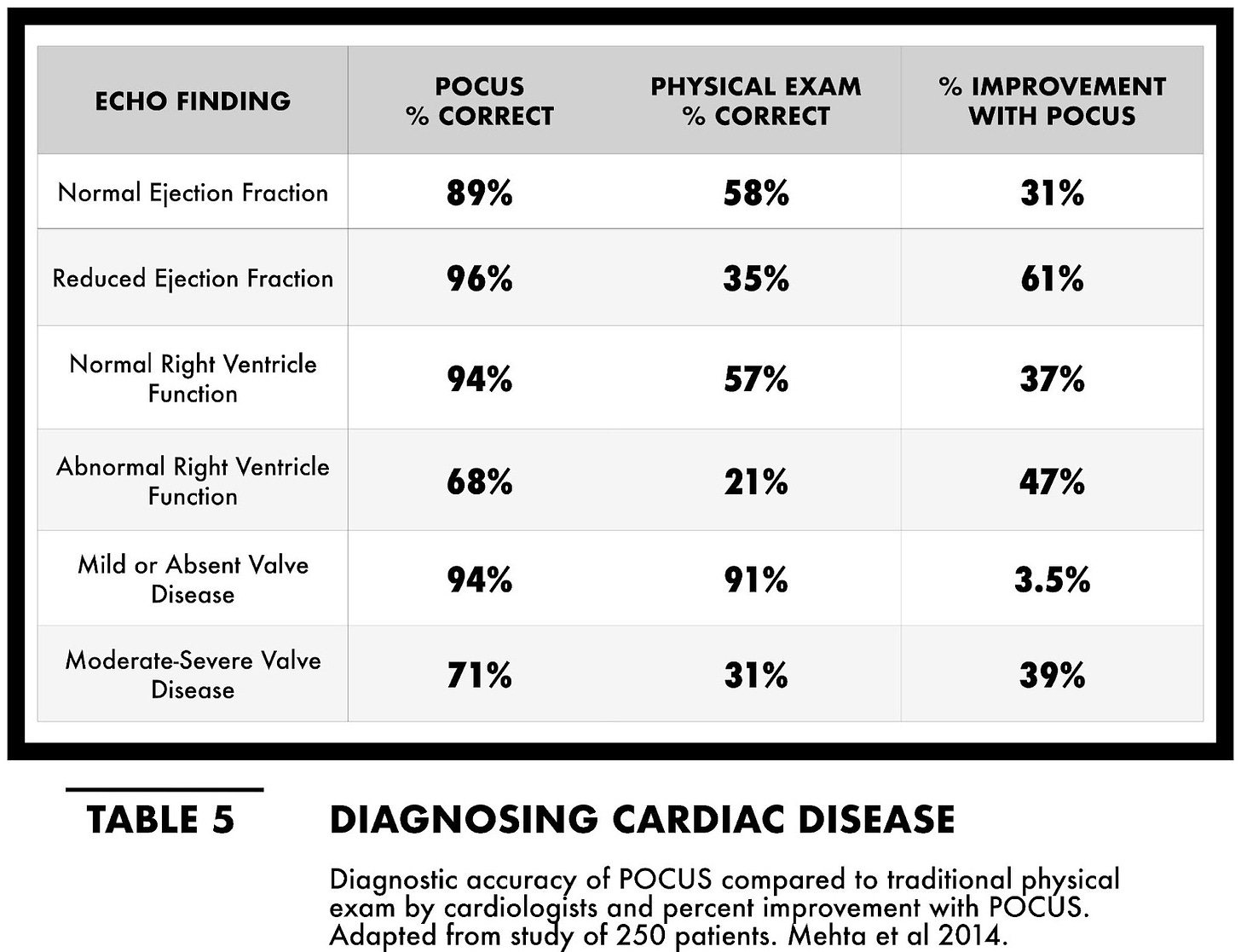
Similar improvement to the physical exam with POCUS has been confirmed by numerous similar studies, solidifying cardiac POCUS as a more accurate and effective method for diagnosing cardiac disease at the bedside.
Redefining “Physical Exam”
In the POCUS Era, murmurs can be seen, not just heard. A reduced ejection fraction is detected, not inferred, and the presence of a pericardial effusion is confirmed or denied, not hypothesized. In a 200 year span, the weeks-long, meticulous auscultation-to-autopsy strategy Drs. Laennec or Hope had to employ to understand cardiac disease can now be achieved with far greater accuracy in just a few minutes, with a dollop of gel and a well-positioned ultrasound probe.
What excitement and disbelief might strike Dr. Hope today were he able to look inside his patients while still alive and see a diseased heart, beating in real-time; and what disgust or bewilderment he may feel toward those who scoff at such potential, blinded by their dogmatic defense of a 200-year-old tool.
With multiple pocket-sized, increasingly affordable ultrasound probes on the market, it is time to expand the classical limitations of what exactly a ‘physical exam’ is and incorporate cardiac POCUS, building upon what Dr. Reolandt advocated more than four decades ago.
—
This was an excerpt from The POCUS Manifesto: Expanding the Limits of our Physical Exam with Point-of-care Ultrasound (POCUS). You can buy it on Amazon here.





